Mastering Immune Complexity
Unlocking the Power of Diversity in the Immune System
The human immune system is a marvel of biological diversity. Each person's immune repertoire is shaped by their unique genetic background, environmental exposures, and life experiences. This diversity is what makes immunotherapy so promising - and so challenging. When we can harness an individual's immune system to fight disease, the results can be transformative. But predicting how a patient will respond to immunotherapy has remained one of medicine's greatest puzzles.
The Complexity of Studying the Immune System
Traditional approaches to studying the immune system have relied on reductionist methods - isolating single cells, studying individual pathways, and focusing on one target at a time. While these methods have yielded important insights, they miss the forest for the trees. The immune system doesn't work in isolation. It's a complex network of interactions between dozens of cell types, each responding to signals from its neighbors and the broader tissue environment. A T cell's behavior depends on the dendritic cells that activated it, the cytokines in its environment, and the tumor cells it encounters.
Why Modern Tools Aren't Enough
Recent advances in single-cell sequencing have given us unprecedented views into cellular diversity. We can now measure the expression of thousands of genes in individual cells. But having data and understanding biology are not the same thing. Current computational models treat cells as independent units, missing the multicellular interactions that define immune function. Even the most sophisticated machine learning models struggle to capture how immune cells work together as a system.
The Dream of Simulating Biology
What if we could simulate the immune system? Not just model it abstractly, but create a digital twin that captures the multicellular interactions, the dynamic responses to perturbations, and the patient-specific variations that determine treatment outcomes? This has been a long-standing dream in immunology and computational biology. But the mathematical and computational challenges have been formidable. Previous attempts have been limited by their inability to scale to realistic tissue sizes while maintaining biological fidelity.
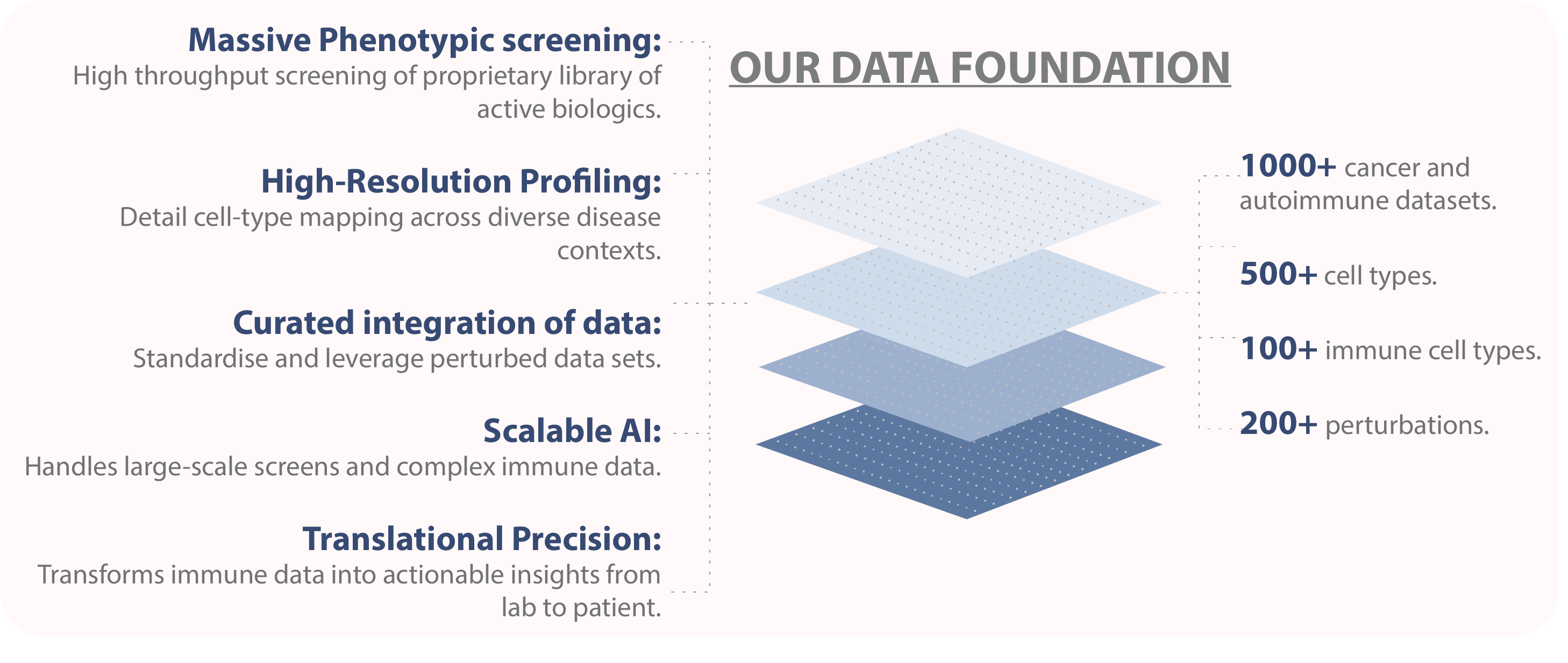
AnuBio's Game-Changing Approach
AnuBio has developed a fundamentally new approach that makes this dream a reality. Our platform combines two breakthrough technologies: PHENOMS, our foundational model trained on over 60 million single cells, and TRAILBLAZER, our multicellular perturbation prediction system. Together, they form a closed-loop system that can predict how the immune system will respond to therapeutic perturbations at the patient level.
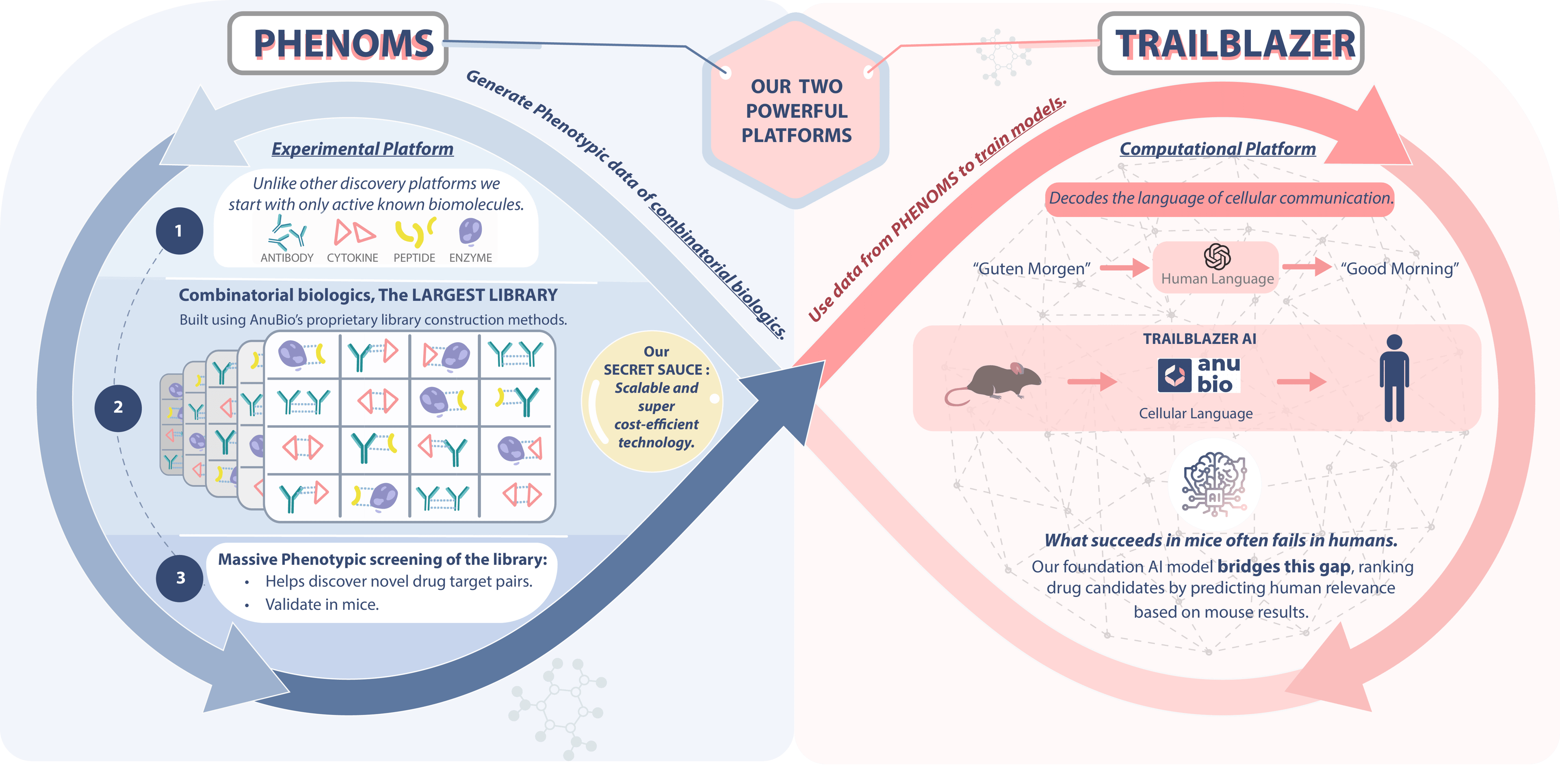
How TRAILBLAZER Sees the Immune System
TRAILBLAZER is built on a novel architecture that natively models multicellular interactions. Unlike previous approaches that had quadratic time complexity - making them impractical for realistic tissue simulations - TRAILBLAZER achieves linear time complexity while maintaining system-level context. This breakthrough allows us to simulate hundreds of thousands of cells interacting in real time, capturing the emergent behaviors that arise from multicellular dynamics. The model doesn't just predict what individual cells will do - it predicts how the entire immune ecosystem will respond.
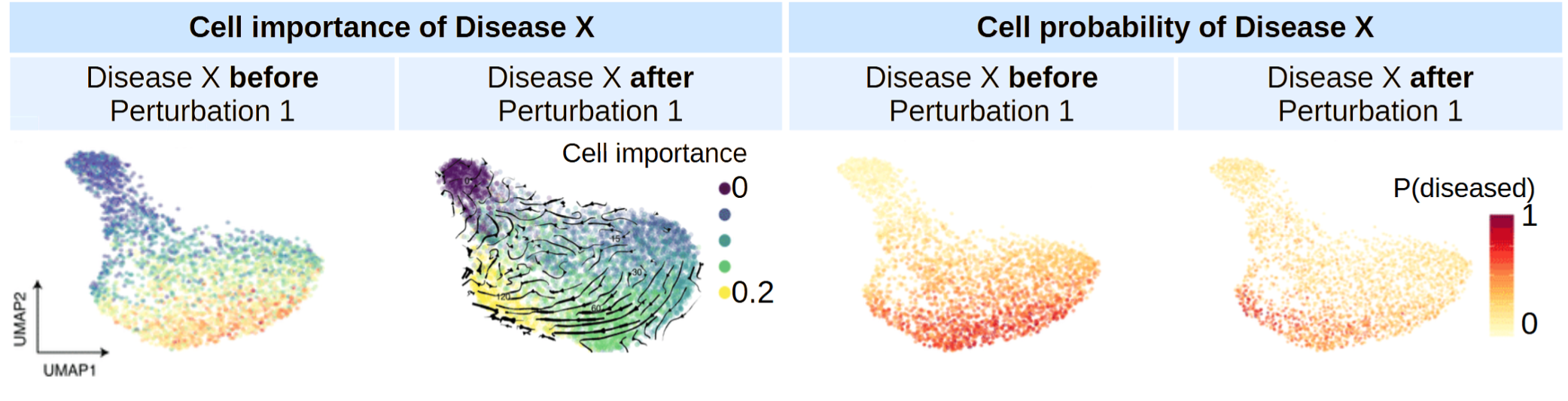
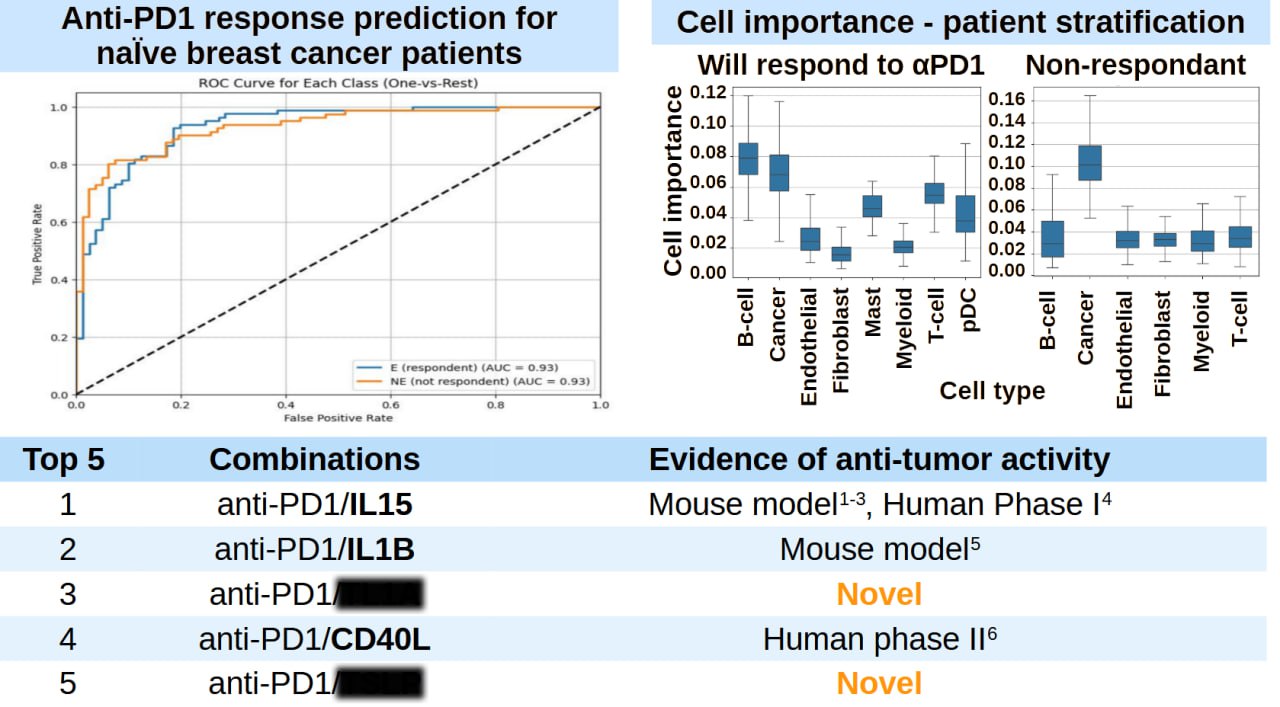
A Case Study: Enhancing Anti-PD1 Responses
In a recent validation study, we applied TRAILBLAZER to one of the most important questions in cancer immunotherapy: how to improve responses to anti-PD1 checkpoint inhibitors. Using data from breast cancer patients, we created digital twins that captured their baseline immune states. TRAILBLAZER predicted combination strategies that would enhance anti-PD1 efficacy. When we validated these predictions against clinical outcomes, the model achieved 85% accuracy in predicting patient responses. More importantly, it identified combination strategies that would have been missed by traditional target-based approaches.
A Vision for Personalized Medicine
This is just the beginning. As we accumulate more single-cell data and refine our models, TRAILBLAZER will become increasingly precise in its predictions. We envision a future where every patient receives a personalized treatment plan based on their unique immune profile. Where clinical trials are enriched with patients most likely to respond. Where combination therapies are designed by AI to maximize synergy while minimizing toxicity. The immune system's complexity, once an insurmountable barrier, is becoming our greatest asset in the fight against disease.
